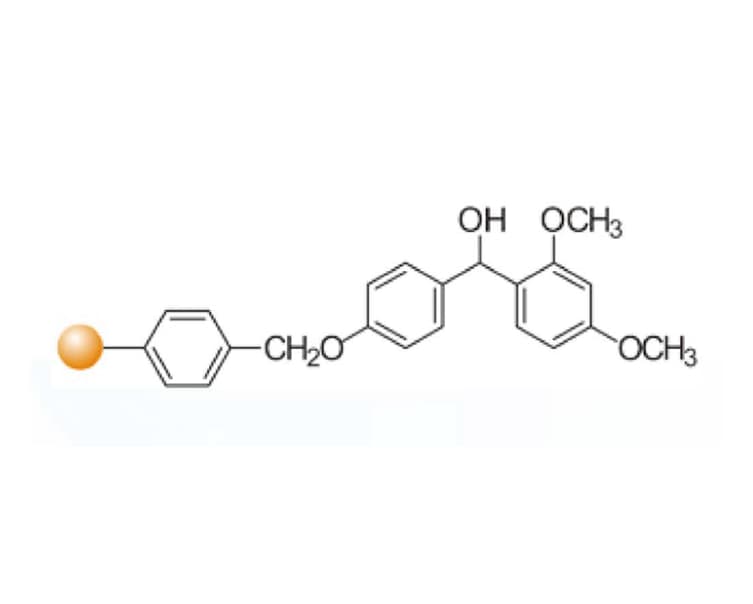
Rink acid resin is a highly acid-labile resin that can undergo cleavage under mild conditions such as 10% acetic acid. However, due to the extreme sensitivity of this resin to acid, the coupling reaction should be carried out under alkaline conditions to prevent the loss of peptide chains. Although the Rink technology is limited to the preparation of amides and carboxylic acids, the use of Rink chloride allows various nucleophiles with a wide range of functional groups, such as amines, alcohols, and thiols, to be attached to the resin and released under mild conditions. Breakthrough of traditional limitations:
Carboxylic acid and amide synthesis: Direct synthesis of C-terminal carboxylic acid (acidification after cleavage) or amide (amination before cleavage) via Rink acid resin. Phenolic directed synthesis: Utilizing Rink trifluoroacetate resin to achieve mild removal and purification of phenolic hydroxyl protecting groups (such as Boc-phenol). Heterocyclic chemistry applications: Purine compounds: Solid-phase synthesis of guanine/adenine derivatives using Rink resin, followed by mild release after constructing glycosidic bonds via the Mitsunobu reaction.
产品编号 Product No. | 交联度(%) %DVB | 粒径(目) ParticleSize(mesh) | 基团负载量(mmol/g) | 膨胀体积SwellingVolum e(inDCM,ml/g) |
| HXDD011B0103 | 1% | 100-200 | 0.3-0.5 0.5-0.7 0.7-0.9 0.9-1.2 | 8-10 |
| HXDD011B0203 | 2% | 100-200 | 6-8 | |
| HXDD011B0104 | 1% | 200-400 | 8-10 | |
| HXDD011B0204 | 2% | 200-400 | 6-8 | |